The novelty of the SARS-CoV-2 coronavirus led to a concerted effort by the global research community to better understand how the virus operates at the molecular level. The highlighted research here suggested answers to questions such as how the SARS-CoV-2 may have evolved from animals to humans, how to improve the potency of peptide vaccines, examining the role of T-cells in immunity, using T-cell receptor sequencing to predict disease severity, and simulating the mechanism by which the virus docks with the body’s receptors.

Loss of glycosylation ability in Spike N370 glycoprotein found to be an important evolutionary event for the enhanced infectivity of SARS-CoV-2
The SARS-CoV-2 coronavirus may have spread from animals such as bats and pangolins to humans. But the molecular changes that enabled this host expansion remain unclear. This study examines the role of the SARS-CoV-2 spike (S) glycoprotein, which mediates viral entry by binding to the host receptor and fusing viral and cellular membranes. The research pinpoints a mutation in SARS-CoV-2 that causes the spike N370 glycoprotein to lose the ability to perform a process involved in receptor-binding called glycosylation. This was likely an important evolutionary event enabling a higher capacity for infection of humans.
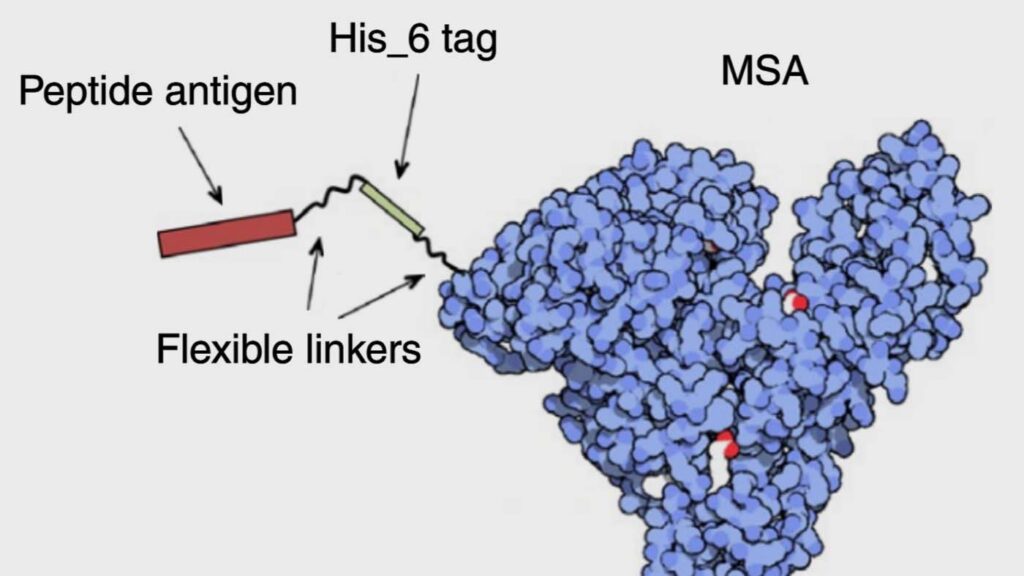
Enhancing the potency of peptide vaccines by fusing the peptides’ epitopes to carrier proteins
The potency of peptide-based vaccines is limited by poor transport to lymph nodes following injection. This research illustrates a method for improving antigen delivery by fusing the portion of the peptide that is capable of stimulating an immune response, the epitope, to carrier proteins. Doing so makes pharmacokinetic “tuning” possible; for instance, the research showed that the carrier protein transthyretin simultaneously optimized three factors involved in antigen uptake and protection of antigen payloads. This method can increase vaccine potency by up to 90-fold, and maximize the responses to viral antigens, tumor-associated antigens, oncofetal antigens and shared neoantigens.

Longitudinal analysis of T cell receptor (TCR) repertoires reveals shared patterns of antigen-specific response to SARS-CoV-2 infection
T cells play a prominent role in the immune response to viral diseases, but their role in subsequent immunity to SARS-CoV-2 infection remains poorly understood. This research studied the assortments (repertoires) of TCRs in an Italian population following a SARS-CoV-2 outbreak. An individual’s TCR repertoire changes as immunity is developed following exposure. Samples taken at 2, 9 and 15 months found elevated levels of TCRs associated with SARS-CoV-2, demonstrating the T cells’ central role in mounting a persistent immune defense against SARS-CoV-2.

T-cell receptor (TCR) sequencing identifies prior SARS-CoV-2 infection and correlates with neutralizing antibody titers and disease severity
The blood levels of neutralizing antibody titers (nAb) closely correlate with the protection provided by an effective vaccination. But nAb assays are challenging to perform at a large scale. This research instead applied a TCR sequencing assay on a standard blood sample to assess T-cell response to SARS-CoV-2 infection. It found that the magnitude of the SARS-CoV-2-specific T-cell response strongly correlates with nAb titer, as well as with clinical indicators of disease severity including hospitalization, fever, or difficulty breathing, thus demonstrating the utility of a TCR-based assay.
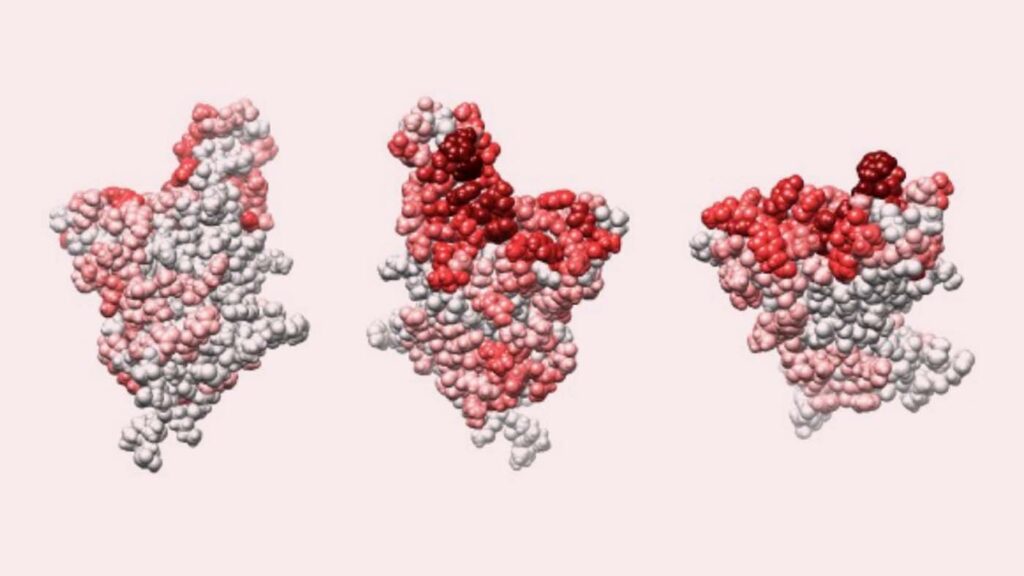
Risk of rapid evolutionary escape from biomedical interventions targeting SARS-CoV-2 spike protein
The spike protein receptor-binding domain (RBD) of SARS-CoV-2 is the molecular target for many vaccines and antibody-based prophylactics for COVID-19. But a narrow molecular focus could simplify viral immune evasion. This research examines how easily the virus escapes neutralizing antibodies (nAbs) that target the spike RBD by combining an analysis of the RBD structure-function with an evolutionary modeling framework. The modeling suggests that SARS-CoV-2 mutants are expected to exist in high numbers due to neutral genetic variation, and consequently resistance can develop quickly -and repeatedly- under positive selection.
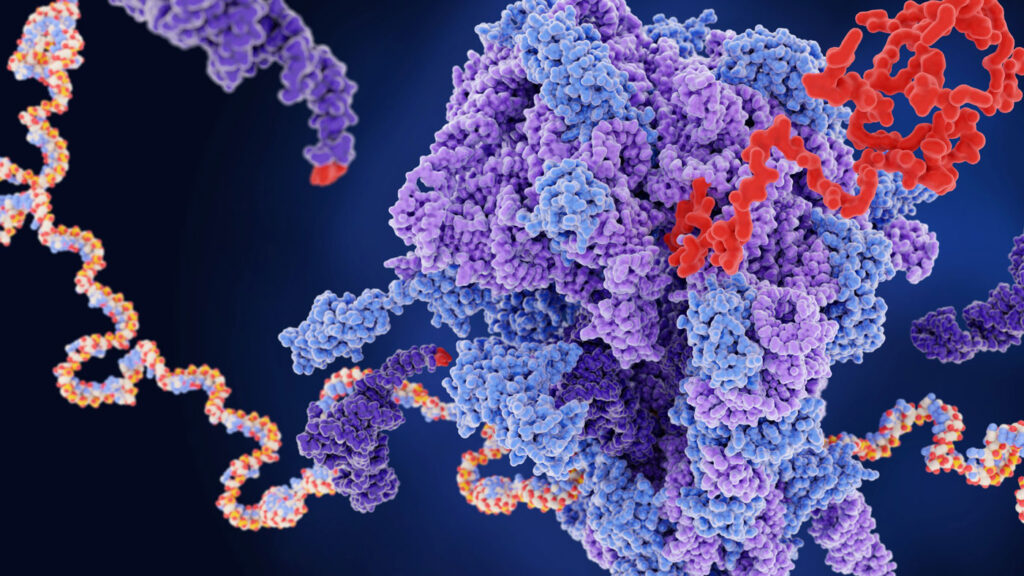
A “wedge” model using molecular dynamics simulation that shows how the SARS-CoV-2 spike protein’s N-terminal domain regulates the motions of the receptor-binding domain
Two key segments of the SARS-CoV-2 spike (S) protein are the receptor-binding domain (RBD) and neighboring N-terminal domain (NTD). The RBD allows the S protein to dock to a body’s receptors to gain entry into cells and cause infection. This research applied a molecular dynamics simulation to the S protein to model the movements of these domains. It revealed that the NTD acts as a “wedge” to prohibit the RBD’s movements, and it occasionally moves out to allow the RBD to tilt downward. This NTD “wedge” model suggests that the NTD–RBD interface should become a potential drug target.

